December 29th, 2013 §
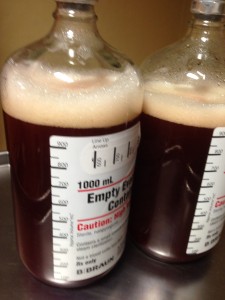 No news isn’t always good news. A lot has been happening in the last few days. On Tuesday they successfully drained 2 liters from my left pleural space after the unfortunate failed attempt last Friday. There is still about one liter remaining in there (the doctor can only safely remove about 1.5 liters at a time). Think about that: two liters. The size of a soda bottle… a few pounds. From one side. It’s astounding how much space and weight that is. Those are the actual bottles of fluid in the photo.
No news isn’t always good news. A lot has been happening in the last few days. On Tuesday they successfully drained 2 liters from my left pleural space after the unfortunate failed attempt last Friday. There is still about one liter remaining in there (the doctor can only safely remove about 1.5 liters at a time). Think about that: two liters. The size of a soda bottle… a few pounds. From one side. It’s astounding how much space and weight that is. Those are the actual bottles of fluid in the photo.
Everyone kept telling me how great I would feel after the thoracentesis. I did feel like I was breathing easier, but I didn’t feel great. In fact, I had quite a bit of pain. They checked for an air leak by x-ray as they always do after this procedure. There were no visible complications. But there was still tremendous pain starting that day and increasing into the next. It was more than the pain from the reinflation of a lung that had been compressed by half its size. But exactly what it was we weren’t sure.
By Thursday when I had to go back to Sloan-Kettering for my PET scan, echocardiogram, and EKG I was in agony. One of the doctors I met with could see how much pain I was in and prescribed some narcotics. I can’t take anti-inflammatories because I am having a liver biopsy next week and they put patients at risk for bleeding. I am so grateful a doctor seeing me about paperwork reached out to offer assistance and relief.
My father met me in the city. After I completed the required tests he drove me home. I can’t think of anyone better qualified to keep an eye on me than a retired cardiothoracic surgeon. Unfortunately, the pain continued. In all likelihood it is a combination of the cancer causing inflammation in the pleura and some nerve inflammation now that some of that fluid is gone and the lung is reinflated. It’s really all conjecture. We will see if the scans and tests reveal any additional information.
Oxycodone couldn’t keep the pain under control so yesterday I had to combine it with a Fentanyl patch. This duo is definitely helping me more; I’m still in pain but it’s more manageable today (Sunday). Each of these patches lasts for 72 hours and will deliver a constant stream of medication to me. I am pretty much bedridden.
It’s looking like Tuesday (New Year’s Eve) will unfortunately be a tough day. I was already scheduled to have a liver biopsy (This is not to check for malignancies; we already know there are metastases here. This procedure will be for repeated genomic sequencing analysis and other testing to make the best choices about chemotherapy and future clinical trials). Now, given all of the events this week, the team has decided that I need to have a PleurX catheter put in. This is a drainage system that will allow me to remove excess pleural fluid at home rather than having to have it tapped with a needle each time it builds up. The catheter will stay in place only as long as I need it, which will be as soon as treatment starts working and the fluid stops being produced in such copious amounts. I assume this will be at least a few weeks.
Having these two procedures and pain control issues means I will need to stay overnight in the hospital for observation. It’s amazing the lengths some people will go to to be in NYC for New Year’s Eve fireworks, isn’t it? Maybe Anderson Cooper will swing by while I’m recovering.
Once we see the results of all of the tests I’ve had and have these two procedures behind me, we will re-assess the plan to start the second clinical trial on Thursday. The most important immediate tasks at this point are to treat the two symptoms and get my breathing and pain under control. Immediately after that we go to work on the metastases to stop the problems at their source.
Thank you for all of the support I’ve had over the last few days including local friends who have been so kind and some friends who came from quite a distance to keep me company this weekend. I’m just putting one foot in front of the other and doing everything I can to gather all of the information I need to make a decision about what the best next step is.
Of course, as much as I can, I’ll keep you posted. Twitter is always the best way to keep up to date on daily happenings. It’s a lot easier for me to get quick bursts of info out in that medium. So many have asked: I have all of the help that I need and respectfully ask for no visitors or gifts during this time.
I would like to wish you all a happy and healthy 2014 if I don’t post again here before we all ring it in.
December 21st, 2013 §
 When I was growing up and there was a particularly tough day my mom would use the expression, “Mama said there’d be days like this.”
When I was growing up and there was a particularly tough day my mom would use the expression, “Mama said there’d be days like this.”
Yesterday was one of those days.
My husband and I left the house before dawn. At about 8 AM I started my appointments. First was a physical exam with vitals taken and a review of symptoms. I met a new oncologist who was filling in while so many of the doctors were away (Friday of a holiday getaway week). We arranged this match because she is the Principal Investigator on a new clinical trial the team has been discussing as a good fit for me. As regular readers know, in my last post I explained that my cancer has progressed in some spots (stable in others) so we need to try to find something better now.
We reviewed my scan results and what she and my regular onocologist think we should do next. A slot in a new trial opened this week and it seems to be a reasonable next step to try another non-chemotherapy method. I’m going to save the details of that study for a later post but for those of you who know and understand the jargon, this one involves a Novartis drug called LEE-011 which is a CDK 4/6 inhibitor in combination with an anti-hormonal agent. This clinical trial is what I’m signing up for next.
The protocol for this drug is very challenging. It involves many trips to the city, especially in the first month where it will be once or twice a week, sometimes just for a blood draw. Some of those visits will be 8 hour sessions where blood is taken a few times to check drug levels in the blood before and after taking the pills. None of the blood draws can be done near my house since the conditions and testing all must be carefully controlled as part of the study. I’ll enumerate side effects and other details in a later post but this one looks to affect me more in daily functioning than I’ve had to deal with in the last few months. I’m definitely nervous.
We talked for a long time about the study. I had already gotten word from my oncologist about her own recommendation. I signed consent forms. I scheduled the necessary tests. They require a CT scan (I just did one, though it falls 2 days outside the testing window. We should be able to get an exemption for that so that I don’t need to redo it). I will need a PET scan, likely need a repeat bone scan, an echocardiogram (this drug can have cardiac side effects), an electrocardiogram, blood test, urine test and, (rats!) a liver biopsy. All of these must be completed in the next two weeks during my “washout period” (interval of time where you are not taking any chemotherapy agents and so you are starting with a clean slate to measure effects in a new clinical trial). Of course, the holidays are not an easy time to accomplish all of these.
I then went to get chest x-rays to monitor my pleural effusion (fluid in the sac around my lungs that makes breathing difficult). After those x-rays and fasting until almost noon I had 40 minutes to finally grab a quick bite. Then I went to the main hospital to meet with a pulmonary physician to decide what to do about the pleural effusion and find out how bad it actually is.
I had a full medical history and symptom assessment with a nurse and then met my new pulmonary doctor. He told me that my left lung is compressed to about 50% of its usual size from the fluid that is there. He estimated 1.5 liters of fluid have accumulated. He said it “layered” on x-ray which means it’s still flowing and therefore would be easier to extract. We decided to do a procedure called a thoracentesis to drain it. One of my morning blood tests that had to do with clotting had come back high, something that would mean we couldn’t do the test. We figured out it had been drawn from my port, which should never be done for clotting tests because they use an anti-clotting liquid called Heparin to flush the port each time and that would lead to inaccurate results.
Through a lab snafu it took 2.5 hours to get the new results rather than 40 minutes. By the time we finally had the all-clear to proceed it was about 5 PM. I still had barely eaten or had anything to drink. For the draining they sit you on a table with your feet dangling and have you bend over a stand for support. It is very much like having an epidural placed. They use local anesthetic to numb the skin and then stick a needle between your ribs, insert a catheter and vacuum extract the fluid (which should take about 10-15 minutes). The doctor got the catheter in, got a small sample of fluid going and…
My blood pressure plummeted. My heart rate dropped. I got hot and woozy and clammy. Yup, I passed out. They had to remove the drain and get me on oxygen and lay me down. I revived quickly, but we could not continue. So, I have the pain from the needle in my back but nothing to show for it. I will now have to go back on Christmas Eve to try again (fully hydrated and fed, hopefully this will be the key). I was obviously disappointed and emotional by this point. I had to go get the X-ray you need to confirm that no damage was done by the needle, and we made a train that got us home at 8 PM. It was a long exhausting day without too much to show for it in terms of relief.
It’s hard not to feel defeated on some of these long days when it just seems the mountain is so big to climb. Right now we are making a change to try to get better results in controlling cancer progression. I won’t be able to to travel for the holidays anyway, but now I will be spending time in hospitals rather than resting at home and taking a break. Cancer doesn’t give a damn about Christmas. Or families. Or anything that matters to me. But my doctors do. And they continue to show caring and concern and work so hard to try to make things better. Without that help and support this would be so much harder. Even when mistakes happen (and yesterday there were quite a few with blood draws and lab tests and so on), every doctor apologized. I definitely shed tears many times yesterday out of frustration, which doesn’t happen too often.
As I waited for my results I watched the office staff exchanging gifts, talking about holiday parties and Christmas cookies. One by one they packed up their belongings and turned out the desk lights. I was the last patient left in that department. That was hard. But I also know that I got to walk out of the hospital last night. I still got to go home and sleep in my own bed. And when I got home I made it just in time to hug my children and see Tristan’s artwork, all sparkly and smile-inducing. He asked me at bedtime why I couldn’t go on vacation with the rest of the family again this year. I explained to him that altitude makes it harder for me to breathe. The air is thin, and I would not feel well. I asked if he understood. “Yes, but I am still sad you can’t come.” “Me too, honey. Me too.”
I’m going to need to dig deep over the next 6 weeks. I’m going to need to ask for help with child care and logistics while I’m recovering from procedures and having so many trips to Sloan-Kettering. That’s not easy either. I find it very hard when I feel that I’ve not been strong enough, or that I’ve complained about the way a hard day has gone. I know it’s normal to need to just cry and complain and say to the cold night-time sky, “This isn’t fair. This isn’t how it is supposed to be.” Sometimes you just need to vent, though.
I need to be strong for the next few days and what they will bring. I want to make the holidays joyous for my family to the degree I can. These are the tasks that make me feel like me. The family shopping is done, the teachers’ gifts distributed, the tips for those who help during the year have been handed out, the holiday cards sent. These are the things that I know I could get a “pass” on. But they are the things I value. I will always try to show my gratitude to others.
Yes, Mama said there’d be days like this. But tomorrow is another day. And I don’t lose hope that it will be better.
December 12th, 2013 §
 Monday morning was an icy, rainy mess. I left the house before dawn, hitching a ride into NYC with Clarke as he drove to work. He dropped me right at the hospital because the weather was so nasty. I only had a short wait until the offices opened at 7:30 and started my first meeting around 8:00.
Monday morning was an icy, rainy mess. I left the house before dawn, hitching a ride into NYC with Clarke as he drove to work. He dropped me right at the hospital because the weather was so nasty. I only had a short wait until the offices opened at 7:30 and started my first meeting around 8:00.
Before each meeting with the Principal Investigator on the trial (an oncologist) I usually meet with a research nurse. On Monday I had the added pleasure of meeting with the Fellow assigned to this trial. She is a fabulous doctor: thorough, curious, caring. We spent more than 45 minutes going over a checklist of symptoms we must review at each appointment. I need to answer if I am having any of those symptoms, describe them in detail, and rate if they are better or worse than at the last visit. Then we discuss what to do to help alleviate the symptoms. Certain medications are allowed and others are not because they could conflict with the investigational drug.
At this visit my main issues were muscle pain in my back, fatigue, occasional bone pain in my collarbone, continued loss of appetite (though my weight has stabilized), occasional shortness of breath, dry skin and cracked heels, and some minor GI issues. My blood pressure and heart rate are elevated. My pleural effusion has stayed at the reduced level, fluid continues just in the lower left lung lobe.
In general, I feel quite good on the combination of GDC-0032 and Fulvestrant. I’m quite happy with my current quality of life on the combo especially compared to other options like IV chemo. I hope that it will continue. I have historically tolerated targeted therapies very well. I have not had any issues so far with mouth sores, rashes, or serious GI issues which are some of the more common side effects with the investigational drug.
I was able to ask all of my questions, most having to do with the coming two weeks. Next week is a big milestone: I will have my first CT scan and that will be the basis for determining if I can continue in this clinical trial. At least two radiologists will examine the CT. One radiologist from the hospital will read the images, while a second, separate radiologist assigned to the trial will make his/her own determination as to how much disease there is compared to the baseline CT 7 weeks ago. If the cancer metastases are considered stable or decreased, I will continue in the trial. If the cancer has progressed (grown) by 20% or more, I will need to stop taking the drugs and be removed from this trial. There are defined days that are the days I must have this scan done so that all of us taking part in the trial are assessed at comparable points in treatment.
Next I met with the Principal Investigator on the trial. This is the oncologist who is the point person for the trial and supervises all of the patients in the trial at Memorial Sloan-Kettering. We reviewed how I am doing in general, what the plan is for the coming 2 weeks, and discussed bloodwork. I told her that am anxious to hear how the other people enrolled in the study nationally are doing. Because we all started within a few weeks of each other, there aren’t many reports yet.
Because I have completed my loading doses of Fulvestrant (an extra dose of the drug is required in the first month of treatment), I did not have to get those injections this week. That was a treat. I will now receive the two injections monthly.
My medication diary documenting the time I stop eating each night and the time I take my medication in the morning was checked as were my pill quantities. I scheduled my next visit (adjusted a few days earlier because of holiday schedules there, I have a +/- 3 day window for the appointments now) and headed up to the third floor to have my blood drawn.
My port incisions are healing beautifully. I’ve toyed with posting a photo of what it looks like but haven’t decided about that yet. The nurse and I donned the requisite masks for port access and the blood draw was easy. I was then allowed to take my GDC pills and start the clock on the 60 minutes until I could eat and drink (I must always wait one hour after taking them). I left the hospital about 3 hours after I arrived.
We’ve been watching my tumor markers and aren’t quite sure what to make of them. They’ve been rising a lot in the past month but I am also getting varied results from my two testing sites. Of course, the key piece of data to look at is the scan. But it’s been a challenging few weeks emotionally as I see where the markers are, watch them rise, and wait for the scan to tell me what’s truly going on inside by body. In a few days I’ll have answers. And then I’ll either be continuing on the trial for another two months or moving on to plan B (which my team and I have already identified).
I continually try to bring my focus back to the distinction between worrying and planning. Worrying is anticipatory. The way I look at it is that worrying is spending time thinking about things that may or may not be/come true. Planning is taking strategic action to set things in place and control things that I can control in the midst of so much uncertainty.
Having a backup plan or a next step if the scan brings bad news next week is planning. It means if this trial isn’t working I know what I will do next and make sure those steps are in place so I’m not suddenly reeling and trying to cobble together a plan. But worrying about the results next week won’t do me any good. The cancer is doing what it is doing. These drugs are either working or they are not. And my sadness or frustration about that won’t change the reality of the cellular processes.
And so I have been quieter this week, choosing carefully how to spend my time. I’m searching for joy each and every day and finding beauty in the small moments: our dog Lucy playing in the first snowfall of the season, Christmas shopping with Paige last weekend, puzzling through math homework with Tristan, Colin and I getting haircuts together and going Lego shopping.
We hug a lot.
We say “I love you” a lot.
We always have done this.
But now I hold on for an extra second each time and I squeeze just a little tighter.
………………………………….
For those readers new to my posts about this clinical trial, you can see my reports about Cycle 1, Day 1 here and Cycle 1, Day 16 here and my port placement and Cycle 2, Day 1 here.
November 26th, 2013 §
 On Friday morning after I sent our three children off to school I traveled to Memorial Sloan-Kettering Cancer Center (MSK) to have a port implanted. My first appointment of the day was at 10:15 to have an electrocardiogram (EKG/ECG). MSK requires an EKG on one of their machines within 48 hours of the port procedure. It takes longer to find the bathroom than it does to have this test done. Electronic leads are attached with sticky sensors to specific points all over your body and hooked up to a machine. After about one minute of recording you are done, and the sticky round patches and the attached wires are removed. Easy as can be. The test measures the electrical activity of your heart to make sure it is normal. I won’t go into details on this test because it’s such a piece of cake and so common.
On Friday morning after I sent our three children off to school I traveled to Memorial Sloan-Kettering Cancer Center (MSK) to have a port implanted. My first appointment of the day was at 10:15 to have an electrocardiogram (EKG/ECG). MSK requires an EKG on one of their machines within 48 hours of the port procedure. It takes longer to find the bathroom than it does to have this test done. Electronic leads are attached with sticky sensors to specific points all over your body and hooked up to a machine. After about one minute of recording you are done, and the sticky round patches and the attached wires are removed. Easy as can be. The test measures the electrical activity of your heart to make sure it is normal. I won’t go into details on this test because it’s such a piece of cake and so common.
After that test was complete (by 10:08) I went to a different floor in the same building to the Interventional Radiology department. There, a friendly team checked me in and sent me to the waiting room, a large attractive area brimming with patients and family members. After a little while a woman called my name and I was taken back to be prepped for surgery about 30 minutes before my scheduled 11:15 arrival time. I will say that for every test and procedure I’ve had so far at MSK’s main hospital they have been on early or on time. This has been a pleasant surprise at such a large medical center.
Once we reached a small prep room a very cheerful nurse gave me a garment bag to store my clothes in and I changed into a standard issue hospital gown with a thin navy blue bathrobe to wear over the top. She weighed me, measured my height, and reviewed my medical history. This was the first surgery I had at MSK so I had to review my surgeries and allergies along with current medications, etc. They wanted to do another pregnancy test but I refused on the grounds that I had an oophorectomy and the test was unnecessary. Because the surgery had not been done there they didn’t have the exemption in my file. They confirmed that I had not used any blood thinners (including medications or pain remedies that can act as blood thinners) in the days prior to surgery.
I then met the surgeon for the first time and he and I discussed the precise placement site of the port and also the ramifications of my sensitivity to adhesives. Usually it’s not too much of an issue but for this procedure the surgeons usually close the incisions with Dermabond (skin glue) and my skin cannot tolerate that. Sutures would be used instead.
The surgeon pinched the skin between my collarbone and the top of my breast implant and said that there was enough tissue there to use the preferred port, called a MediPort or PowerPort. Each person has a different amount of fatty tissue in this area, and a mastectomy may affect this as well. Age, body type and other factors can affect which model of port can be used and where it will be positioned. Obviously, children and people using a port for only a short period of time may have different limitations and needs. There were a few times during the day when someone said to me, “You’ll just have this for a while and won’t even know it’s been there after it’s gone.” They assumed that I would only need it temporarily. “It’s here for good,” I said more than once.
There are many different kinds of ports. They have changed a lot over the years. The one I have is quite small and is triangular in shape. This shape is an indicator that the port can be used for injections of contrast dye (these are called “power-injectable”) in addition to being used for blood draws and any future IV chemo. The power-injectable feature means that when I go for CT scans, bone scans, or PET scans, the technician can inject any dye that might be needed for that test into the port rather than having to use an IV line into my hand or arm. This is one reason I decided to get the port. Being in clinical trials now means frequent blood draws and scans.
The nurse started an IV in the holding area and I was wheeled through a maze of hallways until we stopped outside the operating room. I got off the gurney, walked into the OR, and hopped onto the table. After a lot of prep including hooking me up to monitors and draping and cleaning the area, they finally pushed Fentanyl and Versed into my IV. I didn’t actually go to sleep but probably could have. They numbed the two incision areas with local anesthetic and after about 15 minutes including a few periods of tugging and pushing it was over. I’m going to just link to the actual description that MSK gives about the procedure itself. I’m not sure I can explain it any better than they do.
I stayed in the OR for about ten minutes and then was wheeled to a very small private recovery room where I stayed for about an hour. The one surprise is that in the Interventional Radiology department’s recovery area at MSK they do not give you anything to eat or drink after procedures. So I needed to wait until I left to have anything (tip: if you are going to have a port placed, tuck a snack and drink in your bag. After fasting until the procedure you will want something convenient to eat and drink afterwards and radiology departments might not provide them the way that surgical recovery areas often do).
My husband was able to join me in this recovery area after I was settled. A nurse reviewed my discharge instructions. I needed to wait slightly longer to get the incisions wet than usual because I did not have the Dermabond. Usually it’s a 1-2 day wait. I was quite sore immediately after and was glad I had put a cushion and pillow in the car. If your port is put on the right side, as mine is, the passenger side seat belt will not be pleasant so I recommend bringing a padded seatbelt cover or other method of cushioning the strap. I was quite sore for about 24 hours, but quickly that shifted from being generally aware of the pain to being very localized and only when using that arm. That quickly became localized discomfort if touching it. Today (three days later) it’s still sore to the touch but otherwise not bothersome. I did not use any pain medication.
The port is much smaller than I would have thought. It’s placed so low that it won’t be visible in a tank top. The surgeon was very careful to try to pinpoint a location that would be cosmetically most appealing which I appreciated since this will not be temporary. Eventually I will need IV chemo and this will be used for that as well. For now there are two red incisions but I know those will fade. They are far more visible than the port, a bump under the skin the size of a quarter.
I will not be able to use the port for blood draws that I do near my home and many people are not aware that not all phlebotomists can access the port. Only certain people (most often at hospitals and oncology offices) can access the port because you need special training and also special equipment. In addition, if you do not use the port for a period of about 30 days you must go to have it flushed (with saline and Heparin) to prevent clotting. It only takes a minute to do that.
I’m happy to answer any questions that readers have about the port or anything I missed in the description.
…………………………………
Today I was back at MSK for my regular clinic day for Cycle 2, Day 1 of my clinical trial of Genentech GDC-0032 + Faslodex. I met with a nurse first who checked my weight, blood pressure, oxygen saturation, did a physical exam, reviewed my side effects (I won’t go into those in this post). Next I met with the Principal Investigator on the trial who is the one in charge of my care during this time. I gave her the four remaining pills of my 30 day supply (two each of 1 mg and 5 mg capsules of GDC-0032) and signed and turned in my medication log which lists what time I stop eating each night and what time I take my pills each morning. We discussed scheduling for the month which includes my CT scan three weeks from now where we will get our first look at what, if any, effect the drug combination is having on the cancer.
Next I went to the chemo suite to get my Faslodex injections and have my blood drawn as required by the protocol. I had a different nurse this time from the last two times. I told her that I had a new port and asked her to explain the process for the draw now. She warned me it might be sore as she palpated it to find the right spot to insert the needle and also said the actual needle stick might be a bit more painful today depending on the exact location of my incision.
We each needed to put masks on to cover our nose and mouth during the blood draw to minimize the chance of infection. She gently felt for the circular center of the port where the reservoir is and inserted a special needle to access it. It was a bit tender but certainly not at all more painful than my usual stick on the back of my hand. It really was just a second and it was great to know there was no chance of “failure”… blood would flow! And it did. Six tubes were done quickly and then she flushed the port with saline and Heparin to prevent clotting. The needle was removed, a gauze and paper tape bandage applied (no adhesives for me) and then we removed our masks. Easy and no anxiety. These steps must be taken every time.
Next it was time for the two nasty Faslodex injections. As I’ve written before these are two jumbo intramuscular injections, one in each butt cheek. The nurse kept apologizing and saying how she hates to give them because they’re just so big and painful. They are always very sympathetic when administering these. Today’s were painful, I think the worst of the 3 rounds I’ve had. I think it’s just chance about where they hit and also that I’ve had to do them every two weeks this month during the loading phase. Now I will only need them every month so there is longer recovery in between from the soreness and muscle pain. I find that a heating pad is the most comforting way to ease the pain, I’m definitely getting used to it and also knowing what to expect.
Once done I stopped into the pharmacy (right next to the chemo suite) to pick up my next month’s supply of GDC-0032. I needed to wait to take today’s dose until after my bloodwork. I took my pills with a glass of water while still standing in the pharmacy and noted the time in my new medication log. I would now need to wait one hour before eating or drinking (as I always do when I take my dose each morning).
I went down one floor to scheduling, made my next appointments, and was on my way about 2.5 hours after I first walked back to the exam room. I met a friend for breakfast and had that blissful first cup of coffee at precisely the time allowed. I took the train home, and fell asleep on the way.
I’ll be back at MSK in two weeks for an appointment with the doctor and bloodwork. The week after that will be my scan and then I’ll be back again a few days before Christmas to begin Cycle 3 if all is stable.
If my cancer has grown (“progressed”) by 20% or more, however, the drug combination is deemed not working. I will stop the trial (and be dropped from it) and I will need to move on to something else. I’m already at work researching what that next choice should be.
…………………………………….
We are entering a fragile time of year. The holidays are difficult for many people. Some miss loved ones who have died. Some mourn their own lives, no longer what they were. Grief takes many forms. The pressure to create memorable and uplifting occasions can sometimes be oppressive.
Be kind to those who are struggling during this time; physical and mental illnesses can be especially difficult to manage. Understand that happiness and sadness can coexist. Reach out to others if you can.
Find a bit of beauty in the world. Share it. If you can’t find it, create it. Some days this may be hard to do. Persevere.
November 14th, 2013 §
 Tuesday was a full day for me at Memorial Sloan-Kettering for Cycle 1, Day 16 of my current clinical trial.
Tuesday was a full day for me at Memorial Sloan-Kettering for Cycle 1, Day 16 of my current clinical trial.
With snow flakes and rain mixing in the morning rush, I decided to skip the train and go early with my husband as he made his usual morning commute to New York City by car. I knew this would get me to the office quite early. I was the first to arrive in the waiting room and didn’t mind the long wait. It gets very busy and noisy later in the day and I’m always happy to sit in warmth and comfort of a quiet room. I arrived at about 7:20 and my first appointment wasn’t scheduled until 8:30. I was fasting, so I did miss my morning coffee quite a bit while waiting, though, especially as other patients began to arrive and use the coffee machine!
I meet with the research nurse first. She weighs me (down 2 pounds from two weeks ago), takes my blood pressure (oops, very high using the machine, we’ll retake it manually, oh that’s much better, thank you very much), pulse (in the 90s), oxygen saturation (better than last time, now normal).
We review how I am feeling on the Faslodex and GDC-0032 combination. This is the first time I am discussing it with the team since starting the protocol. She goes through a list of symptoms and asks about each one. The list ranges from gastrointestinal issues (very important to monitor on this investigational drug) to neuropathy to dizziness to fatigue to rash. I am pleased to report that I feel quite good on the combo. I experienced a few days of hot flashes and night sweats from the first injections of Faslodex two weeks ago, I tell her, but that was it. From the GDC I really only have had appetite loss, metallic taste in my mouth, a bit of muscle pain, and slight fatigue as the most noticeable issues. In general, we both agree, I am tolerating it very well.
The biggest change I’ve experienced in the last two weeks is a drastic change in my breathing– for the better. I’m quite sure, I tell her, that my pleural effusion (excess fluid in the lining around the lungs) is almost resolved. This is great news and I am anxious to find out if my doctor hears the change. I also tell her that after much thought I have decided to have a port placed to make my blood draws and tracer injections for CT and bone scans easier.
Next I meet with my own oncologist. Usually I would meet with the Principal Investigator for this appointment. She was not in the office on the usual days this week, though, and I must have my check-ins on certain days in the cycle. I was told about the switch in schedules well in advance. The plan for the long term anyway is that I will transition back to my own oncologist for monitoring. The two doctors work closely together and there is no problem in doing this.
I haven’t seen my oncologist in what seems like a while (probably about one month) and we are happy to be “reunited” for this appointment. We review how I am doing clinically. She also gives me a physical exam and listens to my lungs. Yes, she agrees, there is a big change for the better. We discuss the port, and she orders the needed additional bloodwork for it.
We talk about the trial and other topics. As always, she asks about my family and writing. I tell her about the song I wrote and promise to send her a link to it. I tell her I have been invited to come talk to the class of Fellows at Sloan-Kettering in a few weeks about caring for the metastatic breast cancer patient. I am truly excited about this invitation. There isn’t anything I can think of that is more important than talking to young physicians about ways to make doctors and patients partners in care in light of a metastatic cancer diagnosis. I’ll share more about that visit as the time approaches.
Next I do scheduling for the next appointment in 2 weeks. That will be a much shorter day than today. Each appointment in the first 8 weeks on the protocol has different elements. Today will be a long day. Next time will just be a visit with the Principal Investigator and a blood draw.
 Next I head upstairs to the chemo suite where they have a room waiting for me. They have already called downstairs while I am doing scheduling to tell me which room to go to so I don’t even need to check in. The nurse I had last time comes in to the chemo room. I immediately tell her that she did a wonderful job with the two huge injections of Faslodex last time. I tell her the slow injection rate seems to have worked; I was sore for days (expected) but no bruising or welts. I always try to give as many compliments as I can; I think these oncology jobs must be very stressful and I would bet the complaints come often. If possible, I try to find something good and comment on it. Everyone likes being appreciated.
Next I head upstairs to the chemo suite where they have a room waiting for me. They have already called downstairs while I am doing scheduling to tell me which room to go to so I don’t even need to check in. The nurse I had last time comes in to the chemo room. I immediately tell her that she did a wonderful job with the two huge injections of Faslodex last time. I tell her the slow injection rate seems to have worked; I was sore for days (expected) but no bruising or welts. I always try to give as many compliments as I can; I think these oncology jobs must be very stressful and I would bet the complaints come often. If possible, I try to find something good and comment on it. Everyone likes being appreciated.
Today I will need to have my blood drawn, take my GDC-0032 pills, wait four hours, and have my blood drawn again. I must always wait one hour after taking the pills to eat. By the time I do all of this today it will be noon. The nurse gets a vein in my wrist easily for the required 12 vials and when we see it’s a good flow we agree to leave the line in and try to use it four hours from now and avoid another stick. I’m reactive to adhesive so she wraps me up carefully and I slide my sweater sleeve back down.
I take my pills with a full glass of water and document the time in the medication diary that I am required to keep. Then it’s time to drop my trousers for the two big Faslodex injections. I am not too nervous about this part now that I’ve done it once already. I will get these today, again in two weeks, and then monthly thereafter. I have mastered the art of muscle relaxation for these shots and use a trick of putting all of my weight on one foot while they inject the opposite side. Again the shots are uncomfortable because the viscous liquid is getting rammed into a muscle. But she does it slowly again as she is supposed to, and we’re done quickly. I still am self-conscious as the lightly-frosted sliding glass doors are all that stands between my tush and the hallway, but I’m already over that. I think the idea of facing the wall and clutching a table as needles are jabbed in my backside is probably more laughable.
I put on my coat, knowing the worst is over for the day and go meet a friend for lunch after waiting the required time to eat.
Later, I return directly to the same room I was previously in at the appointed time. The line still in my wrist yields a good flow, a few more tubes are drawn, and we’re done for the day.
Seven hours after first entering the building I am free to go. I still have two more weeks’ worth of the GDC-0032 in my bag and so I do not need to visit the pharmacy. At the next visit my medication log will be checked and my remaining pills will be counted and confiscated before I am given another 30 day supply.
…….
Over the last two days I have ended up needing two more blood draws, met with my local oncologist, and got my monthly Xgeva shot. After 7 needles in 3 days I’m glad to now have a few days off. I ended up needing to repeat one of the tests required for the port placement procedure (Tests related to clotting are routine before any surgical procedure. One of these was slightly elevated and I needed to repeat it. The second test came back just fine).
I also had my tumor markers done. This blood test is not a part of the clinical trial requirements. I’ve decided to watch these to see what they do while taking the drug. I’m sure some people would not choose to do this. This week’s test showed the numbers were quite elevated over previous weeks. We don’t know what that means. I have no increased pain, none of my other bloodwork shows any reason for concern. The numbers sometimes rise on new therapy before they drop. My breathing is significantly improved. So, we focus on the clinical signs and will just have to see what the markers do. I wish they had dropped, but I’ve seen the imperfect nature of this test time and again. I am able to realize this is only one piece of the puzzle right now. It doesn’t affect anything about my treatment plan. A reminder: in this trial success is measured by doing CT scans at varying intervals. My first one will be five weeks from now, at the seven week mark. Only then is a determination made about whether it is working, which would be disease stability or reduction. If the overall “quantity” of cancer is seen as growing by 20% or more, I would discontinue the trial and need to move to something else; it would be considered ineffective for me.
I was sore far more quickly after the Faslodex injections this time even though they weren’t any more painful at administration time. By the time I was on the train back home in the late afternoon I was hurting. For three days now I’ve had a heating pad on as much as possible. It just feels good. The seat heater in my car is getting a workout too. I know it will only be another day or two so I am not too bothered.
I don’t yet have a date scheduled for the port placement. That could be as soon as next week. I also have meetings with my cardiologist (these inhibitors can drastically change lipid levels. My cholesterol and triglycerides on my previous drug, Afinitor, went quite high. Thankfully they are coming down rapidly now but we still need to watch them carefully.) and my endocrinologist (my thyroid levels are now abnormal again. I have Hashimoto’s Disease which is usually very easy to manage but chemo does sometimes cause changes). I’m not in a huge rush to get the port, I just would like to have it in by the time I need to do the next round of scans.
I’m glad to finally be done for the week with appointments, hard to believe the weekend is almost here. I will see what next week brings and then it will be time to be back at MSK on the 25th of November for Cycle 2, day 1.
October 30th, 2013 §
 There is so much to say about the start of the phase 2 clinical trial earlier this week. I think it’s important to be as complete as I can on the main parts. I really want readers to get a sense of what it’s like to go through this and also what to expect if they decide to enter a trial themselves.
There is so much to say about the start of the phase 2 clinical trial earlier this week. I think it’s important to be as complete as I can on the main parts. I really want readers to get a sense of what it’s like to go through this and also what to expect if they decide to enter a trial themselves.
That said, it is obvious to me (and hopefully to you) that my experience is very unique. I have no earthly clue what other trials are like. I just want to make all of the disclaimers that I think you all know already: this is my experience only. If that helps in some way, great. But it can’t possibly tell you what another clinical trial might be like.
I still think it’s valuable. And I know I would have wanted to read posts like these a few weeks ago when I was signing up for the trial. So that’s my guide: if I think it would have helped me, I’m going to share it.
Twitter friends have been telling me that they are interested in five main topics: 1) what is the science behind this drug? 2) what does the treatment consist of/logistics 3) how did I choose this trial out of the ones available 4) side effects physically 5) effects emotionally. The last two will obviously be the ones we follow over time. I won’t be able to address all of those topics here but I’m getting a good jump on them.
This post is long. I’ve opted to just publish it and not divide it up. If you want to read it in chunks you can decide how to divide it up. If you want to skim the science parts, you’ll still have my report of my day at the end. I look forward to hearing your questions and comments. If you have questions I will try to answer them.
First, a bit about the drugs and the science behind them. My trial has me taking 6 mg of an investigational drug called GDC-0032 made by Genentech every morning. I receive two injections of a drug called Faslodex every month, with an extra dose halfway through the first month.
It is important to understand that these particular drugs for metastatic breast cancer are not traditional (cytotoxic) chemotherapy drugs. What I mean by that is that most people think about chemo as being drugs you receive, most often via IV, that makes you feel rotten and your hair fall out. That’s the type most people are familiar with and that class of drugs includes what I had when I had treatment for stage II breast cancer in 2007 (Adriamycin, Cytoxan (least creatively-named drug of all), and Taxol).
Those drugs are cytotoxic (cyto= cells, toxic= poisonous). So, the drugs kill the cancer cells but they kill other cells too. That’s why your hair falls out, you feel sick, your blood counts drop and a host of other issues.
With my kind of cancer (estrogen receptor positive, progesterone receptor positive, HER2 negative) there are other types of drugs to use to try to slow the cancer’s growth down. This is not the case for all types of breast cancer. With some you can only use traditional chemotherapy. In addition, and most importantly for this trial, my cancer shows a mutation in the Pi3k pathway. I know this is getting very science-y. But I’ll try to explain the rationale for this drug.
Many people with my kind of breast cancer (and other types of cancers, we’re learning) show a mutation in this pathway. You might have heard of genomic sequencing. It is testing the tissue of your cancer to see if your particular cancer has any mutations in its coding that facilitate the cancer’s growth (I’m oversimplifying here). This pathway, which is called the Pi3K/AKT/mTOR pathway, can become overactive and drive the cancer’s growth.
I have one mutation in the Pi3k section of the pathway. There are many forms of mutations in the Pi3k pathway. There can be other mutations in other areas as well. Mine is called a Pi3k-alpha mutation.
So what the investigational drug is targeted to do (hence the term “targeted therapy”) is to block this Pi3k/AKT/mTOR pathway that has been over-activated, potentially by this mutation.
This all sounds great. But it’s not so easy. It isn’t as easy as “find the mutation, create the drug, block the pathway, cancer goes away.” We don’t have indications it will ever be like that. The signaling pathways of cancer are highly complex, variable at any given point, and also change over time.
In addition, not everyone with the same mutation responds to the same drug. And combinations of drug seem to work better. Think about doing clinical trials of endless permutations of drug combinations with different mutations, different cancers, in different bodies… well, this is why science seems to move at lightning speed but our advances in treatment just don’t mirror that in all cancers. We have no way at the moment to predict what the best course of treatment is for any individual person. For now, you throw the pasta at the wall and see if it sticks. Unfortunately, our lives are the test cases.
To return to the science, I’ve already tried an mTOR inhibitor in my last treatment phase. That was called Afinitor (combined with another drug called Aromasin). But that targeted a different end of the pathway, and not the mutation. The problem with all of these right now is that the cancer figures out a way around the blockages. It develops feedback loops. If you’ll allow me to anthropomorphize cancer for a minute, it says, “Hey, okay, so you want to block the road? I’ll just detour and still get the end point. I’ll get fuel to the cancer somehow. If you block me, I’ll just keep finding a new way to deliver the goods.” And that’s why metastatic breast cancer is incurable. It keeps finding a way to find fuel and becomes resistant to each thing you throw at it. I became resistant to that Aromasin/Afinitor combination after about six months.
In terms of side effects, unlike traditional IV chemo, with this investigational drug you don’t “feel rotten” right after treatment. It can take weeks and potentially months for side effects (especially some of the serious ones) to take hold. So that’s one way this differs from what people might think. My phone has been buzzing non-stop since Monday: “How do you feel? How do you feel?” I will have some effects from the injections (which are hormonal agents, this one is an estrogen receptor agonist) quickly. I already have some of those. Other side effects from the pills (the GDC-0032) will come later.
So… here we go.
Monday was Cycle 1, Day 1.
If you are interested, my trial protocol is here (I am in the phase II group). This tells you exactly what this study is. If you want to read a bit about the early results of this drug in phase 1 trials you can see that here.
On day 1 I received my GDC-0032 pills for the month (and took the first dose), received two injections of Fulvestrant (Faslodex), and had about 8 vials of blood drawn.
The logistics of getting to the city were a bit of a challenge this week given fatigue and the lingering pleural effusion. The train, subway, and walk were tough but I always try to push myself. Knowing I’d be stuck inside for a few hours definitely had me enjoying the cool crisp fall air on the walk to the hospital.
Because of train times I arrived one hour early for my appointment. I wasn’t sure if I was doing this blood draw before seeing the doctor or after so I settled in. After only a few minutes I was surprised to be called back to an exam room. There I had a long meeting with research nurse whom I’ve spoken with by phone but not met in person. It was an extremely thorough meeting. She answered questions, reviewed the protocol, went through my current medications again, noted all physical symptoms I’m having now. We also discussed my most recent bloodwork (my lipids changed drastically during the 3 week washout period. My prior chemo had raised my cholesterol significantly. My LDL dropped a whopping 100 points in a two week period once I stopped the old chemo, for example). Blood pressure, pulse, oxygen saturation, height, weight. Other research assistants on the protocol come in and talk to me, discuss things, physical exam, as eventually does the Principal Investigator (the doctor in charge of the trial).
It took about two hours to complete these meetings, exams, tests, questions, medication review, etc. Everyone was very thorough and I was offered every opportunity to ask questions about not only the trial but also about any symptoms I was having and how they could be helped.
They also stressed how important it is to call with any and all side effects. As the more serious side effects become more possible/likely, it’s important to report any issues right away so they can be managed before they get too serious. Communication is key in clinical trials. I’ll talk about the side effects more in the future.
For some questions about side effects we needed to refer to the protocol of the trial (Can I get radiation to a bone if my bone pain continues: Yes, but not within the first two weeks of the trial start. Can I get the fluid around my lung tapped if it becomes too troublesome: Yes, at any time).
We also discussed a port for my blood draws/access for radioactive dye injections for CT and bone scans that I will need to have done every 8-12 weeks on the trial. I am still undecided about the port. We agreed to see how it goes in the next few weeks with the blood draws. I can only use my right hand which doesn’t allow for easy access or many misses. They tell me there is a “three miss rule.” If they can’t get the blood they need within three tries, they stop. I have a terrible feeling this rule will come in handy.
At the end of the meetings they handed me many sheets of paper.
First, I received a medication diary where I need to document the time I stop eating every night and the time I take my GDC-0032 pills (the “investigational drug”) the next morning. The pills must be taken on an empty stomach one hour before food, with a full glass of water at approximately the same time every day.
Second was a list of drugs and supplements I cannot take while I am on this protocol.
Finally, there was a schedule for the next 8 weeks of what will happen at each appointment, what tests and drugs I will receive, what I need to do to prepare, and what I need to bring to each appointment. The pills are given in quantities of 30 but I will probably be at appointments every 28 days. The surplus pills must be traded in and counted before I can receive the next month’s supply. Though it is only one drug it is dispensed in two capsules, one dark red, one pine green. One is a 5 mg, one is a 1 mg for a total of 6 mg.
I was then sent to do scheduling for my next appointments. I will need to return in two weeks (plus or minus 2 days) to do vital signs, exam, fasting blood draw (but this time it must be done twice: before taking my pills and then again 4 hours after), and my two injections.
I then went to the hospital pharmacy to collect my pills. They are labeled with my name, the drug name, instructions, and so on, just like a regular prescription. The protocol number is labeled on the bottles too. The bottles also say “Cost $0.00” on them.
This drug is provided free of charge to me but my insurance company is billed for the injections of Faslodex, lab work, and all scans. Trials differ in what is covered. In this case, only the investigational drug is provided free of charge. The rest of the expenses including the visits with the doctors are not provided for free. I am fortunate. My insurance will help pay for those things. Your care is not necessarily fully covered when you are in a trial, contrary to popular belief.
The pharmacy cashier placed the two bottles (one bottle of the 5 mg, one bottle of the 1 mg) in a brown paper lunch bag and handed it to me. In that moment I feel like I have something very valuable and secret in my possession, perhaps even magical. I don’t know if these pills will do anything for me. They could do me more harm than good. They could do nothing. But they might buy me time. Those are, for now, mysteries.
The brown paper bag with the drug bottles inside seems very wrong, far too flimsy for the weight of the hope that lies within.
I then left the pharmacy and headed upstairs to the chemo floor where I had my fasting bloodwork done (by now it was past 11 AM and I was glad I started early. I really don’t mind not eating, it’s the lack of coffee that gets me. Also, the longer I wait the harder the blood draw is likely to be).
In this room I will also receive my two injections. As I enter the room, I’m wondering about those and anxious. The number of needles I have in a month is astounding sometimes. I was the girl so afraid of shots as a kid I once ran into the parking lot to try to run away to avoid getting a vaccination at a pediatrician’s visit.
One chemo room is dedicated to this trial. I have gone to the same room each time so far. The trial tech doesn’t even try to draw my blood. He calls in the reinforcements. Eight or so vials of blood are taken by a nurse who goes after my hand vein with a vengeance. Unfortunately I receive a call the next day that two of the vials are unusable (hemolyzed). This happens sometimes when large blood draws require a lot of work (suction) to get. I was able to go locally the next day to have the two vials redrawn.
After the blood draw it was time for the “standard of care” drug. This is part two of my clinical trial protocol. The “standard of care drug” means I am given not just the experimental drug, but also a drug that is a reasonable option for treatment for this stage of my disease.
On its own (“single agent”) Faslodex works for some people but seems to have a better track record when combined with another agent. My trial is one that combines Faslodex injections (standard of care) with the GDC-0032 (the investigational drug). There is no placebo. This is a phase II trial. I will get both. We will see if taking the GDC-0032 provides a better result than the historical success rate of those who have received the Faslodex injections alone. There will be 60 people nationally doing this combo, about 10 of them at Sloan-Kettering. I am the fourth person, I believe, to get started on it (the trial just opened a few weeks ago at Sloan).
Faslodex is given by injection, intramuscularly, in your rear. No fancy fun way to say it. You drop your trousers and they have syringes that are over 4″ long (can’t find any literature that states exact measure. I’m going to ask to measure it after my shots next round. I confess I saw them when we were done and they made me a bit nauseated and I didn’t want to be precise!). The needles are thick because the liquid that has to be inserted is very viscous and doesn’t go into the muscle easily. I was fortunate and the nurse did a great job. I tried to remember the tip to keep your muscles relaxed during an intramuscular injection, but it’s hard when you know the size of the needle that’s taking aim for your ass.
The first injection really wasn’t worse than a regular shot. The second one hurt more than the first but still less than I had expected. I opened my bottles of pills, took out one of each, drank my water as directed. I noted the time in the diary.
At this point I was free to go. The injection sites were not immediately painful and I was sent on my way.
Everything was incredibly efficient and while I was exhausted, I was relieved. I kept thinking to myself: “Once again after three weeks of not being actively treating my cancer because of the mandatory ‘washout’ period, I am doing all I can. Action feels good.”
I celebrated getting through day 1 by having lunch with my friend Julie Klam which was such a luxury after the poking and prodding of the morning. I took the train home and felt a sense of accomplishment.
So, in case it wasn’t clear, my next appointment will be two weeks after my start date (“Cycle 1, day 15 +/- 3 days” in protocol speak.).
I think the word that most defines metastatic breast cancer to me is “uncertainty.” You have to figure out a way to live with it. My coping mechanisms are research and action. I can only hope these will serve me well.
I thank you for your support and encouragement this week.
October 18th, 2013 §
 I confess I have putting off writing this post. After getting results of my scans I needed a few days to just sit with the information. I have also been dealing with some physical issues during my washout period (an interval where you must be chemo-free) before my clinical trial starts on October 28th.
I confess I have putting off writing this post. After getting results of my scans I needed a few days to just sit with the information. I have also been dealing with some physical issues during my washout period (an interval where you must be chemo-free) before my clinical trial starts on October 28th.
I had CT scans of my chest, abdomen, and pelvis. I also had a bone scan of my body. I had bloodwork and an EKG and a urine test. These are all tests that need to be done before I can start the clinical trial and will be used as my baselines. Monitoring will happen at frequent points throughout my treatment. Bone scans and CT scans will be done every 8 weeks for the first 6 months and every 3 months thereafter for as long as I am on the protocol. I’ll write more about the trial and the drugs (fulvestrant and GDC-0032) when I get underway on the 28th. I had a lot of trouble with with the blood portion as I only have my right hand and forearm for blood. It took six needles in that area (which included two blown veins) to get all of the radioactive tracers in and blood out. I will be discussing the option of putting in a port at my next appointment.
So the short story is that results were not as I’d have hoped. I wanted it to be clear that the cancer was still confined to my bones and lymph nodes, even if it were in more bony locations. This, however, is not what it seems to be.
First I need to explain about the tests. I’m going to briefly try to explain why it’s a bit of apples and oranges to compare my test from August with my tests last week and why all of it is a bit jumbled. In August I had a PET scan. In oversimplified terms this test picks up on high activity metabolic areas. So, cancerous tumors (and also some false positive areas on occasion) “light up” because they have a high number of rapidly dividing cells. It measures activity. CT tests, on the other hand, are static. They are like x-rays. They just take a picture without regard to whether the nodule/lump/tumor etc. is malignant. It shows there is something there but can’t necessarily tell you what it is in terms of metastatic breast cancer. CTs are very precise visually. PETs are not necessarily so. CTs in my case can’t tell us for sure if a spot is malignant.
I now have a small spot showing up on my liver that wasn’t visible on the PET in August. So is it a new metastasis? Or is it a benign spot that just didn’t show up on the PET because it’s not cancer? Who knows. We will have to wait and see if it changes when we do the next scans 8 weeks after the trial start (10 weeks from now). There are also nodules in my lungs and pleura. These are potentially (likely) malignant but the ones in my lungs have not changed size in 8 weeks.
I think that’s as much detail as I need to go into. The worst spots in my bones are my T12 vertebrae and my first rib/clavicle area. There are many spots but those are the ones that cause the most pain.
Unfortunately and most significantly at the moment, the scans also showed that the small pleural effusion (fluid in the lining around the lungs) has grown.
So we have many question marks with a few new areas of concern. For now we watch and wait. The pleural effusion will need to be drained if it grows or continues to cause breathing problems. For now any physical exertion gives me trouble. I need rest throughout the day but am trying to conserve energy for activities with the kids.
None of this news changes anything about my planned start of the clinical trial. Whatever these spots are and wherever the metastases are, the drugs I will start in ten days have the best chance to keep the spots stable and eventually perhaps shrink them. Reports from the phase 1 trial participants showed that often there was a stability period of about 8 weeks before some achieved some tumor shrinkage. I have ten more days to go chemo-free before I begin. I am counting the days. That start day is the most important day I have. A few of us in the trial are starting on that day so hopefully I will get to meet some of the other participants. There are 60 nationally in this phase 2 trial, approximately ten people will be at my location. There is relief in knowing there is no placebo in this trial. I may not respond but it won’t be because I received a placebo.
I wish I had better news about the scans but I also am resigned. This is what this disease does. I must do the best I can given my daily symptoms. There are questions marks and only time will give us answers. I try to have as much normalcy as I can, which often just feels like sleepwalking through the days and a bit of play-acting. But it’s important.
I keep coming back to a few lines I wrote recently:
Cellular biology is King.
But paired to that fateful ruler
I shall be an argumentative, rebellious Queen.
Wring the most out of each day.
Find those bits of joy and beauty,
Make sure that what I’m doing isn’t waiting around to die.
For truly, that would be a waste.
I can’t do anything more to change what’s happening inside. I have a plan and I just need the days to go by to get started. My quality of life is suffering at the moment but I hope that this will change. It is a rollercoaster, one I have been strapped inside unwillingly. Some days are good, some days are not. These happen to be some of the icky ones, but as always, I press onward.
Every morning I say to myself:
Find a bit of beauty in the world today. Share it. If you can’t find it, create it. Some days this may be tough to do. Persevere.
September 30th, 2013 §
 Here we are again. A crossroads.
Here we are again. A crossroads.
The third of many we will have.
I’ll give a bit of a recap. I know it gets quite technical but documenting the details is important for those who also have metastatic breast cancer.
It’s almost one year to the day that I was diagnosed with metastatic breast cancer in my bones and lymph nodes. I started on the cytotoxic daily drug Xeloda first (in addition to monthly infusions of Zometa) which gave me six months of success, shrinking the amount of cancer evident on PET scans.
In April 2013 when my tumor markers started rising I started on the hormonal combo of Aromasin and Afinitor (and also switched from Zometa to Xgeva because of difficult side effects I was having). I started on a 5 mg/day dose of Afinitor with a 25 mg dose of the Aromasin. I gradually increased the Afinitor dose to 7.5 mg/day, and then when my markers began to rise again a few weeks ago, I started on 10 mg a day.
It has now been five months on this combo (in all of its different dose strengths) and my markers are rising. This chemo is no longer working. So, it’s time to decide what to do next.
Regular readers will know that this is typical of metastatic breast cancer: some therapies may not work at all while some will work for a while. Ultimately, though, the cancer becomes resistant to each of these treatments and will progress. Sometimes you get a great response and can reduce the amount of cancer that is present. Oftentimes you settle for the success of “stability” where the cancer isn’t reduced, but it also isn’t growing. It’s often referred to as hanging out with “Stable Mable.” This is still a good response in most people’s books.
Especially when your cancer is still confined to your bones and lymph nodes, stability has the added feature of being life-saving: as long as the cancer is in those places it cannot kill you. Only once it metastasizes to organs can breast cancer kill.
So… now it’s time to decide what to try next (this part gets technical). Right now it’s looking like I’m headed to a clinical trial of a drug that has shown promise in breaking the resistance of hormone sensitive breast cancers especially in people like me whose cancer displays a Pi3k mutation (40% of people with ER+ breast cancer have one of these mutations). This experimental drug would be in combination with a drug commonly used for my kind of cancer (Faslodex). I’ve enrolled with the clinical trial team. This is a Phase II trial so I would be guaranteed to get the experimental drug.
What does this mean? In simple English, it means I’m trying a new drug that’s designed to break the resistance of this particular kind of cancer to treatment. It seems to be especially effective for those with the type of cancer mutation that I have (Pi3k-alpha). If this drug makes the cancer susceptible to therapy, then the other drug I’m taking can do its work. This class of drugs is at the forefront of where cancer research trials are right now.
I’ve been pre-screeened and have a few more hurdles to clear: scans, bloodwork, and a three week “washout” period where I must be chemo-free to allow any residual effects of my current treatment to clear. Am I nervous about a period with no safety net? Yes, a bit. But I also am curious how my body will react to having current chemo clear my system. I’ve had a lot of trouble with things like high blood pressure, high cholesterol, rapid heart rate, shortness of breath, and headaches in the past few months. I’m interested to see how many of these will improve.
The new drugs will have their own series of side effects, some of them might be similar and a few may be different and more serious. So this three week period sits okay with me because if my current treatment isn’t doing anything anymore, it’s of no use anyway.
There will be so much to share about being involved in a clinical trial. And I will share that with you, should I make it through the final screening process. I am learning so much as I go and I think there is a real chance here to inform others about the experience. Trials are as varied as can be. Each has criteria that must be met. Sometimes you end up on the good side of these and sometimes you don’t.
My oncologist looked at a host of trials before we chose this one. I was ineligible for many of them because of the drugs I have already tried, my particular features of my cancer, what cities the trials were offered in, when they were enrolling patients, etc. Some were not attractive to me because of the phase of trial that it was (I’ll explain in a separate post the differences between Phase 1, 2 and 3 trials). I am glad to be in a trial where a drug has already passed initial safety standards and will also be given to every patient in the group (there are no placebos in this phase of trial).
It’s important to note that a clinical trial should not necessarily be viewed as a “Hail Mary” meaning it’s the last remaining hope. In reality, my sense is that this is very rarely the case, at least with Phase 2 or 3 trials. They can only establish true efficacy in people who are still at certain phases of the disease process and haven’t had too many chemotherapy agents already.
To me the key was to find a trial that had a good scientific basis for success, was currently enrolling patients and was within commuting distance. In addition, I think it’s best if the standard of care drug is one you would already be considering using at this point in your treatment. There is a database at www.clinicaltrials.gov where you can search for trials.
I have reserved my spot in this trial. If I meet the testing criteria I will be one of 60 patients nationwide on this particular protocol, about 10 at my particular location. I view it as an opportunity. It is well-suited for my cancer at this time in treatment. It’s using the newest thinking in targeted therapy based on genomic analysis.
It’s scary, yes. But if it doesn’t work I still have standby treatments to go to (and other trials by that time that I hopefully would be eligible for). I know there is no cure. But maybe this one will give me a chunk of time.
I won’t go into details here but I know people will ask what’s involved. The treatments will be injections (Faslodex is two big intramuscular injections in the butt every month after doses every 2 weeks the first month) combined with daily oral capsule (the experimental drug). I will also continue to get an Xgeva injection in the arm each month. There will be a strict schedule of fasting bloodwork in NYC every two weeks and scans every 8 weeks to monitor the cancer’s growth. I’ll have to keep a medication diary. I’ll let you know how everything else works once I am underway. It’s a lot of work, especially in the first two months including all of the meetings and tests just to get started.
There are so many questions I know people will have. If you post your questions in the comments I will do my best to answer them in future posts if I can. You may post them anonymously if you like. For now I’m not going to state the name of the drug (actually just a string of letters and numbers) until I’m actually enrolled.
It was quite devastating to get the news that the current chemo isn’t working anymore. It always feels like the rug being pulled out from under me. I always cry. I always feel like I’m falling.
But plans are my safety net. Options are my lifejackets.
I leap from treatment to treatment on my tippy toes, knowing if I place too much weight I will sink.
This is the dance I do now. Forever.
August 24th, 2013 §
I had my PET scan on Thursday morning and received the results yesterday. I suppose the best way to simply summarize it is that it’s a mixed bag.
There is mild progression from my scan in April. This means there are more sites of cancer in my bones that are large enough to be detected on these scans (“hot spots”). One hip, one side of my pelvis, one thigh, more tissue and some nodes in my upper chest are all newly affected. Many ribs and one vertebrae continue to be pretty plagued as well but were already lighting up on previous scans.
That said, it is good news that the metastases are still confined to my bones and nodes (honestly, I can’t believe the things I call “good news” these days!). There is no evidence on the scan that it has spread yet to my liver or lungs.
So the scan is overall worse than April but still better than October in that the spots are in the marrow and not eroding bone itself (the fracture I had in my collarbone has healed and the cancer diminished there). This is good and also has the benefit of keeping me mostly pain-free. My chemo causes muscle inflammation which hurts but it’s surely preferable to bone pain. Most other side effects have been the same as before.
So here is where it gets tricky. And if you haven’t figured it out by now, this whole business is quite complicated and full of guesswork. My tumor markers had been rising and it looked like we would need to move to a different chemo. At that point (about a month ago), we decided to increase my chemo dose and see if we could get a response. After two weeks my markers dropped. That was right before the scan.
So, one takeaway is that we are learning that my bloodwork is quite accurately reflecting the cancer’s response to chemo. This is not the case for everyone.
So the million dollar question is: is the scan reflecting the peak progression this time and now that I am on a higher dose of chemo if I continue to have lower markers will we see the scan reflect this in another few months? That is, is the scan showing the worst it got and now with the recent increase in chemo it is actually on a downward trend? That’s the hope: continue to see response. Only time will tell where on that dynamic process this static measure is.
This information puts me in limbo yet again. But that’s really what this world of metastatic breast cancer is. And you know, I would rather have limbo than definite bad news (again, I chuckle at the straws I grasp at here…).
I won’t go into details because this post is growing long, but I did meet this week to discuss possibly enrolling in a clinical trial at Sloan Kettering. This would be a phase 1 trial and would be a strict chemo protocol. I will do a separate post about trials in the future because I think there is a lot to learn about them and also a lot of misinformation about their use with this disease. Bottom line on this particular trial: it wasn’t one I would definitely jump to enroll in. Some additional time to explore it and other options some more would be nice. So “limboland” helps with that, too.
For the next few days (and likely the next few weeks) I will continue taking the higher dose of chemo daily, monitoring the bloodwork every two weeks. I’m having shortness of breath for the first time this week and that is quite unpleasant. It is a side effect of the chemo.
The results are not what I hoped for. But onward I go.
I deal with what is, I adjust to new information as quickly as I can and forge ahead into the unknown.
I thank you for your care and concern this week. As always, it takes some time for me to gather, interpret, and process the information before coherently getting it out to you all.
 No news isn’t always good news. A lot has been happening in the last few days. On Tuesday they successfully drained 2 liters from my left pleural space after the unfortunate failed attempt last Friday. There is still about one liter remaining in there (the doctor can only safely remove about 1.5 liters at a time). Think about that: two liters. The size of a soda bottle… a few pounds. From one side. It’s astounding how much space and weight that is. Those are the actual bottles of fluid in the photo.
No news isn’t always good news. A lot has been happening in the last few days. On Tuesday they successfully drained 2 liters from my left pleural space after the unfortunate failed attempt last Friday. There is still about one liter remaining in there (the doctor can only safely remove about 1.5 liters at a time). Think about that: two liters. The size of a soda bottle… a few pounds. From one side. It’s astounding how much space and weight that is. Those are the actual bottles of fluid in the photo.








 Link to Twitter
Link to Twitter